The Pathway Collaborative Community
Join a network of your global peers in a collaborative environment that enables you to ask questions, share insights and explore solutions. This community was chartered by the Chief Quality Officer Forum as a way to advance breakthrough solutions throughout the organization and industry.
Here are some examples of what we are up to:
Peer Connect Events
SME Blog Days
½ Day CQO Conference
Chartered Initiatives
Coordinated Responses to FDA Guidances
Member Library Access
Online Discussion Board
Member Directory of Global Peers
This is how professionals at the top of their field stay sharp, stay connected, and make an impact — together.
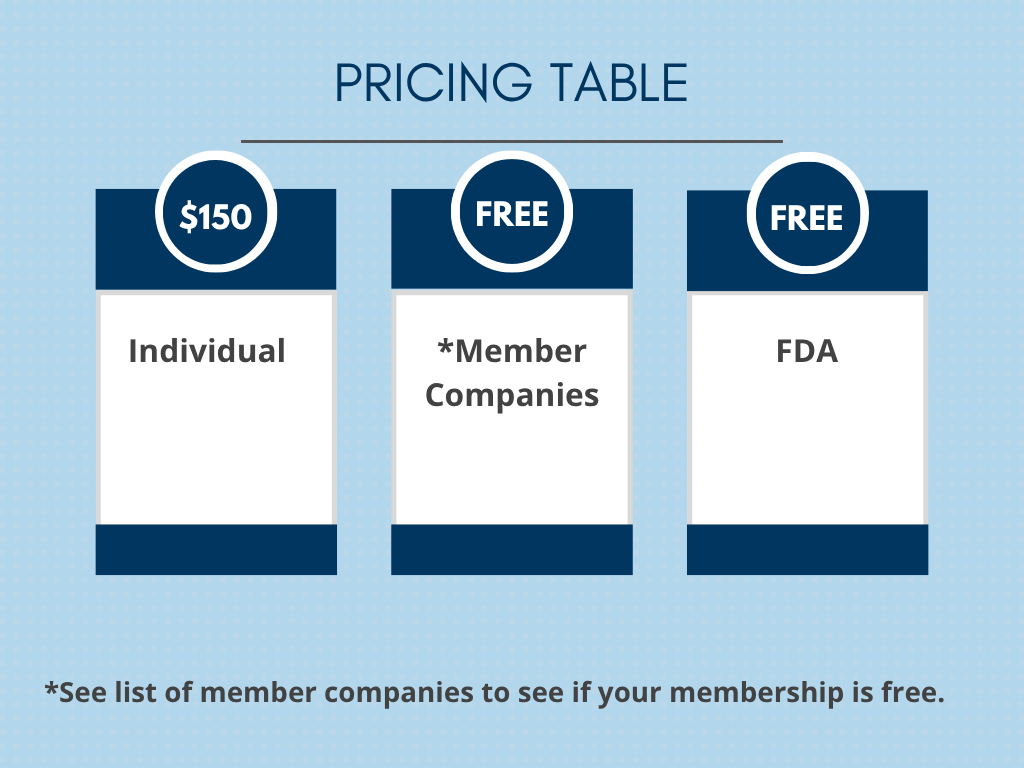
Join Today! Select your membership type below:
*Member Companies
Alkermes
Ascendis
Baxter
Biogen
CSL Behring
Ecolab
Elanco
Fisher & Paykel
Gilead Sciences
Glaukos
Illumina
J&J MedTech
Lonza
MASIMO
Merck
Merz Aesthetics
P&G
Solventum
STERIS
Stryker
Takeda
Thermo Fisher Scientific
Zimmer Biomet
Interested in becoming a Member Company? Please contact Michelle.Rich@Pathway4PH.org
Explore centers aligned with your interest and join their initiatives, including but not limited to:
AI/Digital Center
Human Factors Center
Quality Operations Center
Post-Market Innovation Center
Quality Science Research Center
And more to come!
Purpose of the Community
To proactively foster successful practices gained through a vibrant network of global peers collaboratively addressing contemporary and complex challenges.
In today’s world of constantly evolving innovation and regulatory expectations, industry professionals in the life science industry must stay abreast of these changes and work to advance how their company operates for the betterment of patients around the world. We need to move beyond the “challenges” and into the realm of “solutions”, and at Pathway, we believe we can do this…together.
How our Community Operates
Industry professionals engage in discussions with each other through a facilitated platform that provides consistent opportunities to reach their peers on topics that are most important to them. Discussions often result in identifying initiatives that members are welcome to join.
Each member can:
Join as many Centers in the Forum as desired
Participate in live meetings
Share relevant public news and documents with each other
Invite each other to join initiatives
Present topics of discussion
…And much more!
Examples of How We Engage:
Centers of Collaboration Initiatives. Explore centers aligned with your interest and join their initiatives, including but not limited to:
AI/Digital Center - Good Machine Learning Practices (GMLP) paper to include new developments and Generative AI. Aligning work to support ISO/TR 24971:2020. Conducting AI initiative for Product Quality Reviews with real data.
Human Factors Center - Identifying unique use-related risks that AI and automation bring to products or processes, such as automation bias, complacency, loss of situational awareness, and loss of skill. Outcome includes evaluation methods and tools.
Post-Market Innovation Center - Evaluating and characterizing the post-market data landscape for industry and regulators (e.g., FDABAA-25-00123). Strengthen post-market surveillance and feedback through the total product lifecycle.
Student Research Center - Advancing Scientific Innovation with Quality Science students and Alumni who will answer questions that are challenging industry practice and/or product development. Members will have the opportunity to provide input into research areas.
Quarterly External Intel Meetings. All members are invited to join the quarterly external intelligence meetings, which showcase Subject Matter Experts on topics that are important to the advancement of our industry.
Expert Sessions. Members receive special invitations to meetings with the Chief Quality Officer Forum, and live sessions with experts on critical topics. Unprecedented opportunity to ask questions!
Pathway Forum. This online discussion forum allows members to ask questions of each other, share insights, add upcoming events to their calendar and access shared resources.
Engage with Each Other: Each member can invite other members to join a live meeting they want to host on certain topics, or invite them to participate in industry initiatives.
Check us out!
Check out examples of live meetings, global experts and meaningful discussions you can have access to through the Pathway Collaborative Community [for a complete list of recordings, access our resource library - Resource Library]:
Digital Quality
〰️
Artificial Intelligence
〰️
Annex 1 Changes
〰️
EU MDR/IVDR Revisions
〰️
Data Integrity
〰️
Quality Culture
〰️
Supply Chain Disruption
〰️
Single Use Systems
〰️
Electronic Batch Records
〰️
Workforce Retention
〰️
Metrics that Matter
〰️
Continuous Process Validation
〰️
Global Inspection Readiness
〰️
Quality Productivity
〰️
Diversity and Inclusion
〰️
Digital Quality 〰️ Artificial Intelligence 〰️ Annex 1 Changes 〰️ EU MDR/IVDR Revisions 〰️ Data Integrity 〰️ Quality Culture 〰️ Supply Chain Disruption 〰️ Single Use Systems 〰️ Electronic Batch Records 〰️ Workforce Retention 〰️ Metrics that Matter 〰️ Continuous Process Validation 〰️ Global Inspection Readiness 〰️ Quality Productivity 〰️ Diversity and Inclusion 〰️
Pathway Collaborative Community Leadership
Shannon Hoste is an expert in Quality and Regulatory science that spans years within the FDA and industry. As an engineer, Hoste’s expertise includes Human Factors Engineering, Risk Management, New Product Design Development and Digital Health. As the Pathway Chief Scientific Officer and Associate Professor of the Quality Science Education Program, Hoste has a passion for collaborative engagement and learning.
Marla Phillips, Ph.D. is an expert in Quality Operations from the pharma industry, and has led cross-industry initiatives since 2008. Phillips was a Site Quality Officer for Merck, and then led AI, device and pharma initiatives with global regulators for 14 years through Xavier University. As the founder of Pathway for Patient Health, Phillips leads the Chief Quality Officer forum and developed the Quality Science Education Program, which trains global students to join our industry. Phillips has a passion for making a difference, and looks forward to engaging with you.